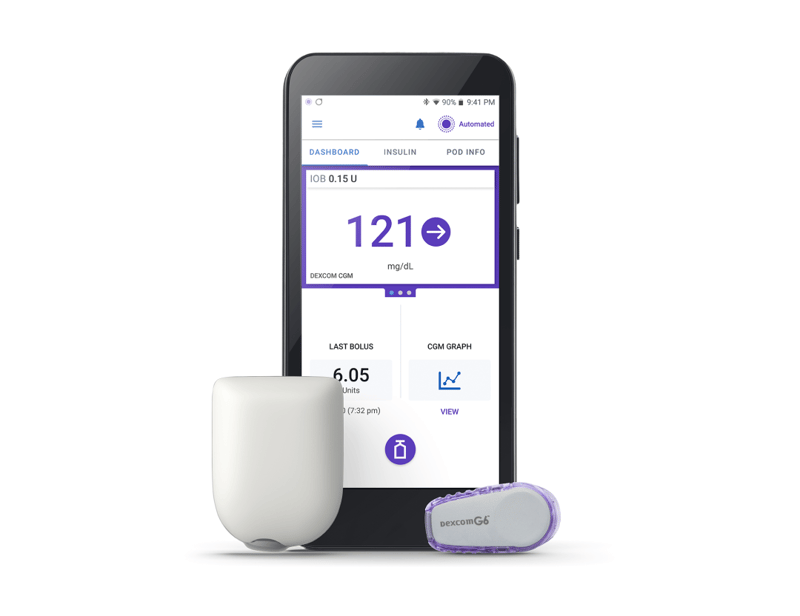
3 Simple Parts

Omnipod 5 controller

Pod

You are viewing the global website


Omnipod 5, the first wearable, tubeless, hybrid closed loop system integrated with Dexcom G6, is now CE marked for people with type 1 diabetes aged 2 years and older.
The Omnipod 5 System works with the Dexcom G6 Continuous Glucose Monitoring System to continuously adapt and automatically deliver basal insulin according to your personal needs.



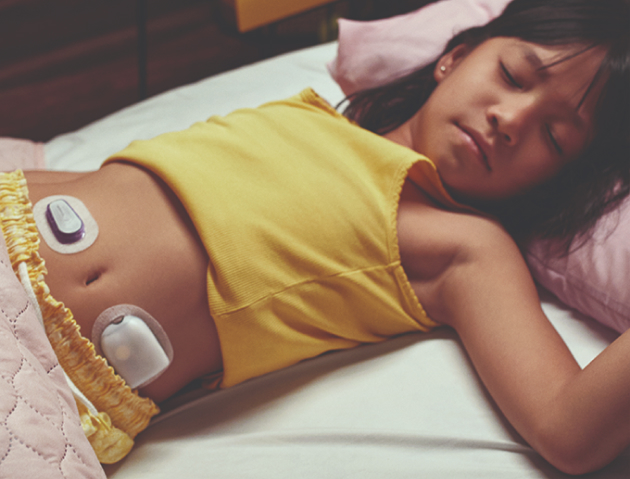
Omnipod 5* with SmartAdjust™ technology automatically increases, decreases, or pauses insulin delivery, every five minutes, to your personal needs which may help prevent highs and lows.
Shortly the latest press release will be available here for download.

Register for the newsletter to receive relevant information and updates on product availability.
Below are some answers to commonly asked questions about Omnipod 5.
The Omnipod® 5 System is the first wearable, on-body, tubeless, hybrid closed loop system integrated with Dexcom G6 approved in Europe for use in people with Type 1 diabetes, aged 2 and above. The Omnipod® 5 System works with the Dexcom G6 Continuous Glucose Monitoring System to continuously adapt and automatically deliver insulin according to your personal needs.
Automated Insulin Delivery (AID) refers to a system that is able to automatically adjust insulin delivery based on glucose readings from an integrated glucose monitor.
Within this there are Hybrid Closed Loop (HCL) systems. These automatically adjust basal insulin based on glucose readings from an integrated glucose monitor, with the user manually delivering bolus insulin to cover meals.
Currently, all available AID systems on the market are HCLs, which mean that there is still a need to manually deliver bolus insulin at mealtimes. Therefore, you may hear these 2 terms used interchangeably
We are delighted to announce that Omnipod 5 has now received CE mark in Europe. However, this is the first step of bringing this product to market. The next key step is to work with authorities to secure access and availability through the healthcare systems. We expect to start launching Omnipod 5 in the first countries in Europe from the middle of 2023 and thereafter on a country by country basis.
At this stage, as we work on getting the product available in your country, we are still working on providing the knowledge to your health care team to be fully prepared at that time.
Register your interest here to be kept up to date on the latest information and product availability in your country.
Choice and access to technology for people living with Type 1 diabetes is of the upmost importance for Nordic Infucare, and so we will continue to supply both Omnipod DASH® and Omnipod 5 systems, recognising the needs and preferences of HCPs and people living with diabetes.
The Omnipod 5 Automated Insulin Delivery System is indicated for use by individuals with type 1 diabetes mellitus in persons 2 years of age and older. The Omnipod 5 System is intended for single patient, home use and requires a prescription.The Omnipod 5 System is compatible with U-100 insulins.
